Structure:
EDTA or ethylenediaminetetraacetic acid is a molecule to create complex metal ions. It is an acid with a polyprotic structure and is constuted of 4 carboxylic acid groups (acidic hydrogens are red) and two amine groups with lone pair electrons (green dots). EDTA is synthesized on an industrial scale from ethylenediamine, formaldehyde, and a source of cyanide (HCN or NaCN).
Polydentate ligand:
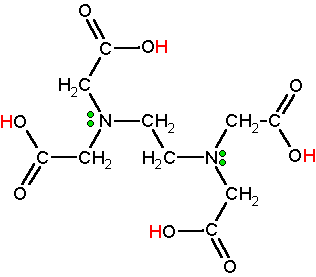
Polydenatate ligands have a range of atoms used to bond to a central metal atom or ion. EDTA contains six donor atoms with separate pairs of electrons that are used to bond to the metal atom or ion and is therefore called a hexadentate ligand.
