Bohr vs Schrodinger model of the atom
| |
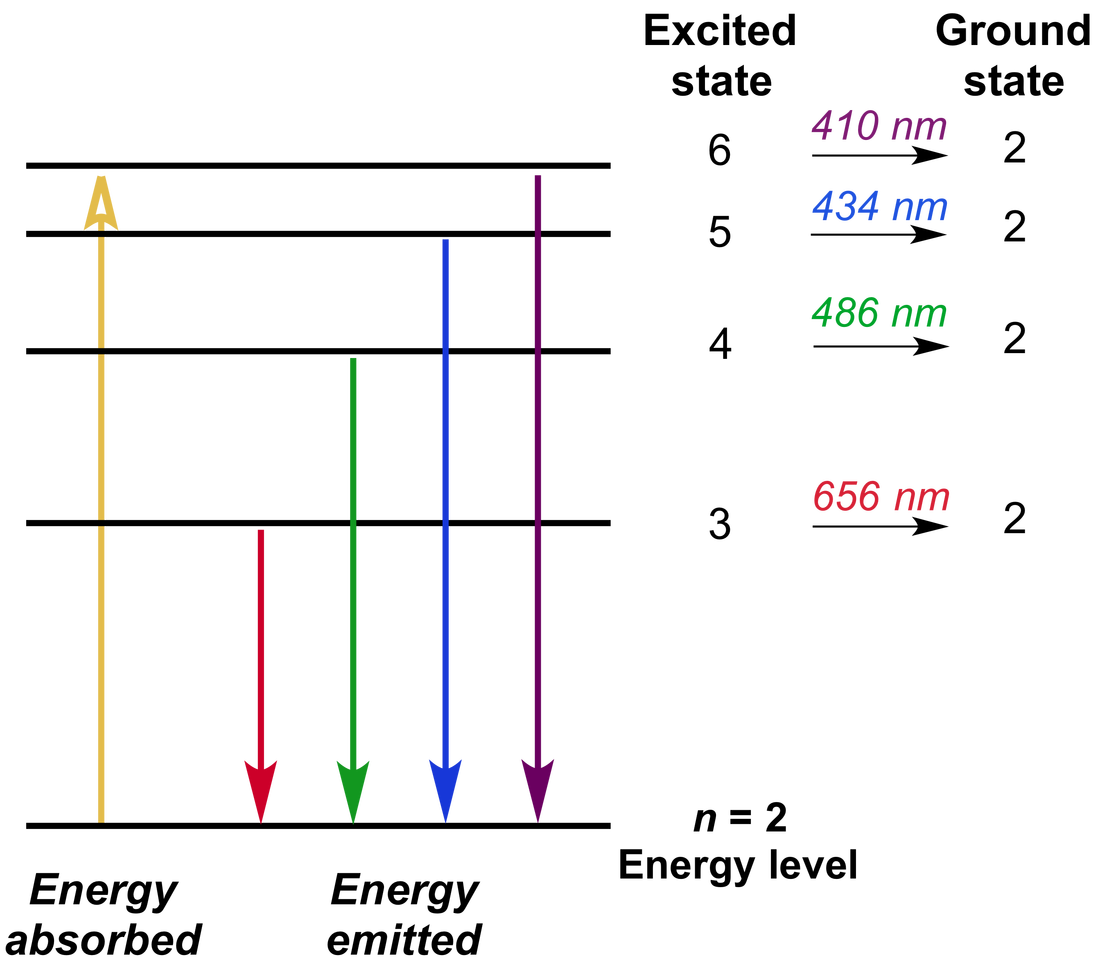
| Bohr's atomic model is based on the concept of electrons moving along paths called shells around the nucleus. The calculation of the energy of an electron in a shell is done using the formula; E(n) = -1/n^2 * 13.6eV Bohr's explanation of the hydrogen spectrum involved electrons absorbing and emitting photons to change energy levels. However this concept did not function in systems containing more than one electron. Bohr's model suggested that electrons absorbed energy in photons which would excite them to a higher energy level if the difference in energy between initial and final levels was equal to the energy of the photon. Changing to a higher energy level would destabalise it causing the emission of a photon to go back to a stable lower energy level. While the Bohr model explained the working of hydrogen and other single electron systems it could not be applied to the spectra of more complex atoms. In addition, there were many unknowns such as the cause of some spectral lines being more intense or the splitting of spectral lines in the presence of a magnetic field known as the Zeeman effect. In the following decades, other scientists like Erwin Schrödinger found electrons to behave like waves as well as particles proving the impossibility of predicting both the position and velocity of a given electron at the same time (Heisenberg's uncertainty principle which contradicts the idea of electron shells and radii from Bohr's model). Thus only the probability of finding electrons can be calculated for a given region surrounding the nucleus. |